Stoyan R. Vezenkov and Violeta R. Manolova
Center for applied neuroscience Vezenkov, BG-1582 Sofia, e-mail: info@vezenkov.com
For citation: Vezenkov, S.R. and Manolova, V.R. (2025) Sensory Disintegration versus Sensory Deprivation: Vestibular-Visual Dynamics and Shared Behavioral Phenotypes in ASD and Early Blindness. Nootism 1 (6), 4-12, https://doi.org/10.64441/nootism.1.6.1
Abstract
This review analyzes the diagnostic and mechanistic intersection between two seemingly opposing developmental profiles: children with Autism Spectrum Disorder (ASD), characterized by atypical, hyper-dominant, or "2D-locked" visual functioning, and children with blindness or severe visual impairment (VI), where visual input is absent and development relies on auditory-tactile strategies. The analysis focuses on the "diagnostic trap" created by shared behavioral phenotypes, including stereotypic movements, spatial insecurity, and difficulties in social referencing. We propose an integrative framework based on multisensory re-weighting among the visual, vestibular, and proprioceptive systems. In typical development, these inputs are balanced to maintain stable posture and social attention. In blindness, the core deficit is Sensory Deprivation, leading to a compensatory re-weighting toward vestibular-proprioceptive channels; here, stereotypies often serve as functional self-calibration ("blindisms").
In contrast, we introduce a model of Sensory Disintegration for ASD, wherein a pathologically dominant visual channel actively suppresses bodily anchors, creating an integrative mismatch. Within this framework, we discuss novel clinical markers – the Screen-Induced Pathological Vestibular Reflex (SIPVR) and Screen-Induced Pathological Eye-Covering Reflex (SIPECR) – which differentiate vestibular suppression (ASD) from vestibular compensation (blindness). The review concludes with a practical framework for differential diagnosis, emphasizing the need to assess sensory integration pathways rather than relying solely on behavioral checklists, and calls for the empirical validation of these proposed markers.
Keywords: ASD; autism; blindness; visual impairment; CVI; visual dominance; multisensory integration; sensory re-weighting; stereotypies; pretend play; ADOS modifications; early screen addiction; SIPVR; SIPECR; cue-dependent behavior; synesthesia
Introduction
Similar behavioral manifestations can be observed both in children with blindness or severe visual impairment and in children diagnosed with Autism Spectrum Disorder (ASD): limited or atypical visual social orienting, difficulties in joint attention, stereotypic movements (rocking, spinning, tapping), rigidity and rituals, and specificities in play and language pragmatics. This phenotypic overlap creates a dual diagnostic risk: (a) over-diagnosis of ASD in children with blindness (when behavior is interpreted through vision-dependent criteria) and (b) missing actual ASD plus visual impairment comorbidity (when symptoms are attributed solely to the lack of vision).
Contemporary reviews emphasize that diagnosis in this population is structurally hindered, as standard "gold standard" instruments include assessments of vision-dependent behaviors (e.g., gaze, facial expression, visual referencing)(Stevenson & Tedone, 2025).
In parallel, evidence is accumulating in autism regarding atypicalities in visual processing (including face and gaze perception, motion and optic flow processing) and, more broadly, differences in multisensory integration and postural control when visual or somatosensory signals are manipulated. There is a marked interest in objects, colors, shapes, and various visual stimulations, rather than in people and social stimuli. Compulsive, seeking behavior for visual stimuli – such as screen stimulation, spinning or illuminated objects, etc. – is observed, alongside a lack of interest in and integration of facial micro-expressions, gestures, gaze, joint attention, and other social contexts. Thus, children with autism acquire "social blindness"; they are blind to social stimuli and communication due to visual hyper-dominance and dependent behavior toward inanimate, artificial visual stimuli – screens, colors, and shapes.
The recently discovered and described pathological reflexes involving the interplay between the vestibular and visual systems in children with autism point toward unraveling the internal mechanisms of their interaction and how this manifests in external stereotypic and rigid behavior.
The aim of this review is to formulate a mechanistic framework that allows for the description of similarities and differences without confounding diagnostic categories. We propose distinguishing between two basic processes:
- Sensory deprivation: absence or minimal visual input (e.g., congenital blindness or severe functional visual impairment).
- Sensory disintegration: presence of visual input, but with impaired reliability or ecological validity and suppression of bodily anchors (e.g., hyper-dominant "2D" visual functioning in ASD).
Rather than asking whether blindness "causes" autism, the more useful question is: how does the brain construct social functioning and a sense of orientation when (a) vision is absent, or (b) vision is present but dominates in a way that disrupts multisensory calibration (Chisari et al., 2024).
Definitions and Scope of the Review
In the present text, the term "visual dominance" is used as a working concept that unifies several frequently described phenomena in autism:
- Atypical orienting of visual attention:g., toward illuminated or spinning objects, colors, and shapes rather than faces or gaze;
- Atypical face and gaze perception: involving networks around the fusiform gyrus/FFA, STS, amygdala, etc.;
- Vulnerability of the dorsal stream/motion processing: optic flow, global motion;
- Potentially different "re-weighting" of sensory channels during posture and movement control (Nomi & Uddin, 2015).
This is not an assertion that "all children with autism have a visual problem," but rather that acquired visual processing and redirected selective attention regarding visual stimuli shape the social-communicative profile.
Under the general term "blindness or severe visual impairment," it is important to distinguish at least two clinically distinct scenarios:
- Peripheral impairments (retina, optic nerve, eyeball), where visual input is limited due to ocular etiology;
- Cerebral Visual Impairment (CVI), where visual pathways and/or cortical processing are affected, and vision may be functionally unpredictable.
This distinction is key because CVI is often accompanied by other neurodevelopmental vulnerabilities ("ocular-plus" profiles) and may carry a higher risk for actual comorbidity with ASD, whereas in isolated peripheral blindness, a significant portion of "autism-like" behaviors reflects adaptation to the lack of vision and peculiarities in the development of orientation (Chung & Son, 2020; Fazzi et al., 2019).
Methodological Note
This text is a narrative (descriptive) review that synthesizes:
- Publications on autism and visual processing (face/gaze/motion, postural control, and sensory re-weighting);
- Publications on development in blindness/congenital visual impairment (social communication, theory of mind, play, stereotypies, and neuroplasticity);
- Publications on the diagnostic assessment of autism in children with visual impairment, including attempts to modify and validate instruments (Stevenson & Tedone, 2025).
Conceptual Framework: The Vision-Vestibular-Proprioception Triad
Spatial orientation and "body awareness" are governed not by a single sense, but through the continuous integration of three inputs:
- Vision – external reference (horizon, optic flow, distances, direction of movement);
- Vestibular system – internal accelerometer (linear and rotational accelerations, gravity, head position);
- Proprioception/Somatosensory system – body map (joints, muscle tone, support, pressure) (Manzari et al., 2022).
The brain combines these signals through dynamic "sensory re-weighting": when one input is unreliable or missing, the weight of the others increases. This process is critical not only for postural control but also for social attention, as stable posture and stable gaze are prerequisites for social referencing, imitation, and joint attention.
This principle is central to the present review for two reasons:
- In blindness: the visual channel cannot calibrate balance and self-motion, which increases the relative weight of vestibular-somatosensory signals and sometimes leads to self-generated rhythmic movements as a regulatory strategy (Fazzi et al., 1999).
- In autism: there is evidence of differences in postural control and sensory integration, especially when visual signals are removed or the reliability of sensory information is modified.
Figure 1 presents three simplified modes of integration that are useful for comparison between typical development, early blindness/severe visual impairment, and an ASD-like phenotype characterized by hyper-dominant visual functioning.
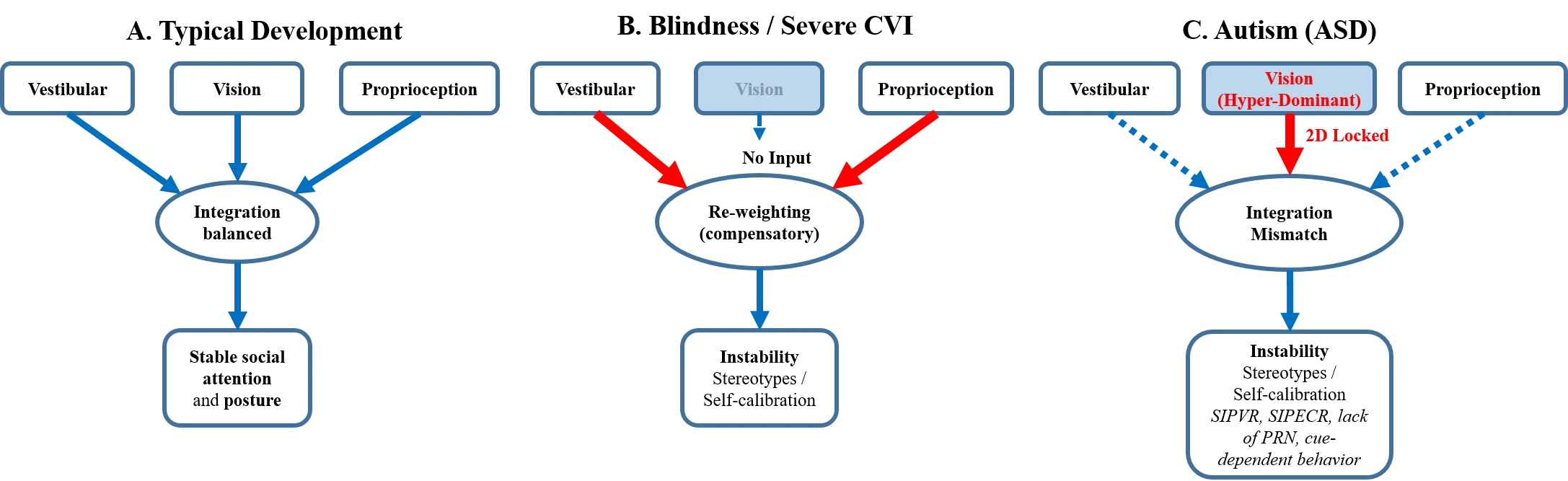
Figure 1. Comparative Model of Sensory Re-weighting and Integration among the Vestibular System, Vision, and Proprioception:
(A) Typical development (balanced integration); (B) Blindness/Severe functional visual impairment (compensatory re-weighting); (C) ASD/Screen-associated phenotype (hyper-dominant "2D" vision and integrative mismatch). Panel (C) highlights proposed clinical markers (SIPVR, SIPECR) and phenomena such as the lack of post-rotatory nystagmus (PRN) and cue-dependent behavior.
Vision as a Scaffold for Social Learning and Postural Stability
In early childhood, vision performs a dual function: (a) structuring the social scene (faces, gaze, facial expressions, gestures, attention orienting) and (b) calibrating spatial stability (horizon, optic flow, predictability of movement). For this reason, impaired visual accessibility or impaired reliability of vision can produce secondary difficulties in social referencing and play that resemble ASD.
Vision is often viewed as the dominant modality for early pre-verbal learning - for orientation toward faces, gaze following, attention synchronization, imitation, and building social predictions. In the context of visual impairment, this raises the question: which social skills are visually dependent, and which can develop supramodally (without visual training) through hearing, touch, and interoception (Begeer et al., 2014).
Important clarification: the lack of vision does not negate social motivation. In congenital blindness, a range of social skills can develop via an alternative pathway through hearing, touch, and interoception (e.g., recognizing emotional states by voice or touch), and occipital areas can reorganize for non-visual functions. Therefore, the phenotypic overlap between blindness and ASD is not evidence of identical etiology; more often, it reflects shared limitations in access to reliable socio-sensory markers.
Several lines of evidence complicate the "simple" model of "no vision → no sociality":
- Spontaneous facial expressions of emotion can manifest similarly in congenitally blind and sighted individuals in naturalistic situations (Matsumoto & Willingham, 2009).
- Facial mimicry (e.g., "smile in the voice") can be observed even in congenitally blind participants, supporting the idea that part of the imitation mechanisms may be modal-general (Arias et al., 2021).
- Neuroimaging and meta-analytical data show significant cross-modal reorganization of occipital areas in early blindness, which become "re-tuned" for auditory-tactile, linguistic, and other functions (Zhang et al., 2019).
This means: blindness does not "produce" autism automatically, but it can create a distinct pathway to social functioning that sometimes resembles an autistic phenotype especially in the early years and with certain etiologies/comorbidities (Fazzi et al., 2019).
Sensory Deprivation: Blindness and Severe Visual Impairment
Early blindness serves as a model for cross-modal plasticity: areas of the occipital cortex can be recruited for auditory and tactile processing and for higher-order functions (language, tactile perception, Braille), a finding supported by meta-analytic and structural data (Zhang et al., 2019). This reorganization is adaptive, but it implies that social learning occurs without the visual "scaffold," which alters the pace and strategy of development.
Social Communication and Social Referencing without Vision
In blindness, social referencing ("what does the situation mean for the other person") occurs without gaze and mimicry cues. Children may seek contact through voice, touch, and proximity, but the pace of automating social rules may be slower, especially in the early years. Consequently, some children display behavioral indicators resembling ASD (e.g., lack of visual joint attention), but in reality, these reflect a different communicative strategy rather than necessarily diminished social motivation.
Data on Theory of Mind (ToM) in blind children are nuanced: some studies show results similar to typically developing children when appropriate instruments are used, while others show small but significant differences or distinct profiles based on etiology (e.g., "ocular" vs. "ocular-plus" blindness, where neurological factors are present) (Begeer et al., 2014). This is important for differentiation from autism: in blindness, there may be an alternative pathway to ToM (more verbal-auditory, less "intuitive-visual") without this being autism.
Play, Symbolization, and Language Pragmatics
Symbolic (pretend) play is highly dependent on the observation and imitation of scenes and roles. In severe visual impairment, the child has fewer visual scenarios for "rehearsal," which can lead to poorer narrativity, more sensory and tactile play, and slower mastery of symbolic substitutes. This has direct implications for differentiation from autism, because "impoverished symbolic play" is a frequent marker for ASD as well – but in blindness, it may be a consequence of limited access to visual-representative scenarios and fewer opportunities for imitative learning.
Similar profiles are described in language pragmatics: structural language may be good, but its use for social purposes may require additional "scaffolding" (verbalization of context, clear rules, role-playing games with tactile supports) (Federici et al., 2025). In a sample of children with congenital visual impairment and normal verbal intelligence, continuing vulnerabilities in social communication and pragmatics are described despite good structural language skills - a profile that may appear autism-like but may have a different causality (Tadić et al., 2010).
Stereotypies and "Blindisms" as Self-Calibration
Stereotypic motor behaviors (rocking, spinning, tapping) are well-described in congenitally blind children and are studied as a phenomenon with various forms and frequencies, including comparisons between "blind-only" children and children with blindness and additional neurodevelopmental impairments (Fazzi et al., 1999). Within the framework of the vision-vestibular-proprioception triad, these movements can be interpreted as self-generated vestibular-proprioceptive signals that support postural calibration and reduce uncertainty stemming from the lack of a visual horizon. Clinically, this means that the presence of stereotypies in blindness is insufficient grounds for ASD; a functional analysis is required (self-regulation/orientation versus rigid models with reduced social motivation).
This represents one of the largest clinical zones of overlap with autism: the same behavioral signal (rocking, spinning, rhythmic movements), but potentially different functions (self-regulation and sensory calibration in blindness; different mechanisms in ASD, including the Restricted and Repetitive Behaviors [RRB] domain).
ASD and Atypical Visual Functioning: From Social Gaze to Posture
The literature on ASD describes variable but often persistent characteristics in visual processing and multisensory integration. In the context of the present review, we use the working concept of "visual dominance/distortion" to unify several phenomena: (a) atypical orienting of visual attention (toward details/non-social objects), (b) distinct strategies for face and gaze processing, (c) potential vulnerability in motion processing (optic flow/dorsal stream), and (d) altered sensory re-weighting in postural control. It is important to emphasize: this does not imply that ASD is a "visual impairment".
Face, Gaze, and Social Perception
The social use of gaze requires rapid decoding of faces, direction of attention, and biological motion. Neurocognitive literature on autism describes differences in face processing and social perception, including the involvement of structures such as the fusiform gyrus/FFA and superior temporal sulcus (STS), which are crucial for gaze, biological motion, and social signals (Nomi & Uddin, 2015). This may manifest as less frequent social referencing, weaker spontaneous imitation, and difficulties in attention coordination (joint attention). Within the framework of "visual dominance," this can be viewed as reduced reliability/priority of social visual signals.
The Dorsal Stream and Motion Processing
Significant literature exists regarding vulnerabilities/anomalies in motion and optic flow processing in autism, often linked to the dorsal visual stream. This has potential consequences for "where/how" orientation and visual-motor integration (Spiteri & Crewther, 2021).
Motion and optic flow processing are critical for the sense of self-motion and for predicting the dynamics of social scenes. Some studies on ASD discuss atypicalities in the dorsal stream ("where" pathway), which has potential implications for visual-motor integration, posture, and navigation in environments with high motion.
Postural Control and Sensory Re-weighting
Postural control is multisensory: when one input (e.g., vision) is uncertain, the brain increases the weight of vestibular and somatosensory signals. In children with autism, differences in postural control have been described, which manifest more strongly under conditions of modified sensory information (e.g., when visual signals are removed). In one clinical study on children with ASD, indicators of weaker postural control and different sway patterns were observed upon removal of visual cues, with discussions pointing to potentially less developed sensory integration for balance (American Psychiatric Association, 2013; Chisari et al., 2024). Within the framework of the present review, this can be interpreted as a different balance of sensory weights or more difficult integration between sensory channels - a mechanism that may phenotypically approach what is seen in deprivation (blindness), but with a different primary causal profile.
Interpretation for the Purpose of Comparison with Blindness:
- In blindness, the issue is a "lack of visual input for calibration";
- In autism, the issue is more often an "atypical use/integration of available input".
Visually-Induced Vestibular Suppression in Children with Autism
Vision in ASD is not absent but pathologically dominant. Conceptually, this can be described as "2D-locked" vision: the visual channel becomes the primary regulator of arousal, attention, and sense of security, while simultaneously suppressing bodily anchors (vestibular and proprioceptive information). The result is an integrative mismatch that can produce an ASD phenotype.
Children and adolescents with ASD had greater postural instability, particularly with eyes closed, compared to neurotypical individuals, regardless of age group. Data suggest impaired sensory information integration in these individuals, but the review did not find any studies that specifically assessed the vestibular system in order to rule out any dysfunction in this system (Helena et al., 2025).
Vestibular dysfunction in ASD includes postural instability, gait dysfunction, and impaired gaze. Untreated vestibular dysfunction in children can lead to delayed milestones such as sitting and walking and poor motor coordination later in life. Histopathological studies have revealed that subjects with ASD have significantly fewer neurons in the auditory hindbrain and surviving neurons are smaller and dysmorphic. These findings are consistent with auditory dysfunction. Further, the cerebellum was one of the first brain structures implicated in ASD and studies have revealed loss of Purkinje cells and the presence of ectopic neurons. Together, these studies suggest that normal auditory and vestibular function play major roles in the development of language and social abilities, and dysfunction in these systems may contribute to the core symptoms of ASD (Mansour et al., 2021).
The following subsections describe proposed clinical markers and phenomena reported in a series of publications from clinical practice. As these data originate from a single clinical center and have not been independently replicated, they should be viewed as hypotheses and clinical proposals for future validation rather than established diagnostic standards.
- The Phenomenon of Absent Nystagmus and "Spinning-Seeking"
In typical development, rapid spinning activates the semicircular canals, leading to dizziness and reflex eye movement (post-rotatory nystagmus) – the brain's attempt to stabilize the image. In children with autism/screen addiction, a paradox is often observed:
- The child seeks intensive spinning, sometimes for hours.
- After stopping, dizziness and nystagmus are absent.
The visual system becomes so dominant ("avian brain" model) that it literally "blocks" the vestibular input. Spinning is a desperate attempt by the body to breach this blockade and generate a physical sense of self, but visual fixation often prevents normal nystagmus.
- Pathological Screen-Induced Reflexes as Markers
Instead of merely "sensory peculiarities," we can now define specific pathological reflexes that distinguish this condition from the norm and from blindness:
- SIPVR (Screen-Induced Pathological Vestibular Reflex): When turned upside down, instead of pleasure or adaptation (as in the norm), the child reacts with panic, rigidity, and an attempt to "anchor". This indicates that three-dimensional space is not integrated; the brain functions in a 2D (screen) mode, where gravity and vestibular orientation are suppressed by static visual dominance. In typically developing children, such a maneuver usually elicits play and laughter or rapid adaptation, whereas in children with ASD, the reaction can be disproportionate, prolonged, and difficult to inhibit (V. Manolova & Vezenkov, 2025b; S. Vezenkov & Manolova, 2025b; S. R. Vezenkov & Manolova, 2025).
- SIPECR (Screen-Induced Pathological Eye-Covering Reflex): This is a key differential marker. Upon covering the eyes (even playfully with hands, a mask, or an object), children with ASD react with extreme panic, hysterical reactions, and in severe cases – aggression or self-injury (S. Vezenkov & Manolova, 2025a). A key feature is that the reaction is sustained as long as the visual occlusion is present and subsides upon restoration of visual input.
Mechanism: Since vision has become the sole channel for security and regulation (hyper-dominance), its removal is experienced as a total loss of control and an existential threat, not simply as darkness (it is not nyctophobia). The constant conflict between the vestibular and visual systems may be overcome through rocking, spinning, rhythmic movements, as well as RRBs (Manolova & Vezenkov, 2025; V. Manolova & Vezenkov, 2025b; S. Vezenkov & Manolova, 2025b, 2025a).
- Synesthesia and Cue-Dependent Behavior (Behavioral Rigidity)
The frequently described "autistic rigidity" (e.g., eating only in front of a screen, using only specific routes) can be explained as induced synesthesia and cue-dependent behavior (V. Manolova & Vezenkov, 2025a). Early audio-visual overstimulation disrupts the process of "pruning" (synaptic pruning), leaving parasitic connections between sensory centers. This leads to conditions where physiological functions (sucking, eating, defecation) are impossible without a specific audio-visual stimulus. This is not stubbornness, but a neurological inability to initiate the program without the external cue.
Comorbidity: Are there "Blind Children with Autism" and When is the Risk Higher?
Yes, co-occurrence is real, but it is heterogeneous and strongly dependent on etiology. A Swedish population-based study of children with congenital/early blindness (born 1988–2008) reports that isolated blindness is uncommon; approximately three-quarters of the children have at least one additional disability, the most common being intellectual disability and ASD. Furthermore, ASD is more frequent in specific etiological groups (e.g., ONH, ROP, LCA, microphthalmia/anophthalmia) (de Verdier, Kim, 2018).
Important clarification: these data do not imply that "blindness causes autism," but rather that there are subgroups in which blindness and ASD coexist.
In broader samples of children with visual impairments, a higher prevalence of ASD is reported compared to the general population, varying according to the type of impairment (e.g., different rates in CVI versus peripheral impairments). In peripheral cases, a diagnostic overlap between symptoms of vision loss and clinical characteristics of ASD is emphasized (Fazzi et al., 2019).
Phenotypic Overlap: Similarities Between Autism and Blindness (and Why They Are Misleading)
Similarities (Descriptive)
(A) Atypical "Eye Contact" / Visual Social Orienting and Difficulties in Joint Attention
- In blindness: Physiological limitation.
- In autism: Vision is present, but social orienting/processing of gaze/faces is often atypical (Zilbovicius et al., 2006).
(B) Stereotypies / Self-Stimulation (Rocking, Spinning, Tapping)
- In blindness: Frequently described motor stereotypies ("blindisms") (Fazzi et al., 1999).
- In autism: Part of the RRB (Restricted and Repetitive Behaviors) domain; motor stereotypies may also be present, but in a different context and with different functional associations.
(C) Difficulties in Symbolic Play
- In blindness: Limited imitative/visual representative learning; data indicating weaker pretend play (Federici et al., 2025).
- In autism: Often limited symbolization, narrativity, and shared play.
(D) Communication Pragmatics
- In congenital visual impairment: Vulnerabilities in social communication and pragmatics have been described, even in the presence of good structural language skills (Tadić et al., 2010).
- In autism: Pragmatic deficit is a core component of the socio-communicative criteria.
(E) Rigidity/Rituals and Context/Environment-Dependent Behavior
Why Similarities Do Not Imply "The Same Thing"
This constitutes the central thesis of the review: the phenotype may overlap, but the etiology and mechanism often differ.
- In blindness: The "social channel" is limited at the input level (lack of visual social signals), but the motivation for interaction may be preserved and manifested through hearing/touch.
- In autism: Difficulties are more often related to the way the brain prioritizes, integrates, and predicts social information, including visual (faces/gaze) and dynamic (motion) information (Nomi & Uddin, 2015).
Key Distinctions for Differential Diagnosis: A Practical Framework
The pivotal distinction lies in whether the limitation occurs at the level of input (absence or severe deficit of vision) or at the level of integration (unreliable or pathologically hyper-dominant vision). Table 1 provides practical guidelines for clinical differentiation.
Table 1. Mechanistic and Clinical Differences between Blindness/Severe VI and ASD (Diagnostic Guidelines)
|
Domain |
Blindness / Severe VI (Visual Impairment) |
ASD |
|
Social Seeking |
Often seeks contact through voice/touch; may utilize alternative strategies. |
Variable; in some cases – reduced spontaneous social orientation. |
|
Stereotypies |
Often self-regulation/calibration under conditions of limited visual input ("blindisms"). |
Often part of RRB (Restricted and Repetitive Behaviors); may also be related to sensory dysregulation. |
|
"Eye Contact" |
Impossible/severely limited (not a diagnostic marker in itself). |
Quality of gaze/social use of gaze is key (when vision is present). |
|
Symbolic Play |
May be delayed/impoverished due to limitations in observation and visual representation. |
Often limited narrativity/plot and reduced social sharing of play. |
|
Assessment with ADOS/ADI‑R |
Standard versions are problematic; adaptations and clinical experience are required. |
Standardly applicable in sighted children. |
|
Vestibular System |
Stimulation seeking ("blindisms") for calibration due to the lack of a visual horizon. |
Suppressed vestibular system due to visual dominance. Post-rotatory nystagmus is often absent during spinning. |
|
Inversion Response (SIPVR) |
Orientation via hearing/touch; panic is rare if security is provided. |
Strongly positive SIPVR: Panic, trembling, and muscular stupor when turned upside down. |
|
Eye-Covering Response (SIPECR) |
Not applicable (no vision) or neutral. |
Strongly positive SIPECR: Extreme panic/aggression upon withdrawal of visual input. |
|
Rigidity / Routines |
Linked to spatial security (need for consistency). |
Overcoming constant conflict between the vestibular and visual systems. Cue-Dependent Behavior: Physiological functions (sleep, food) are locked to specific audio-visual stimuli (synesthetic links). |
Diagnostic Tools: Why Standard "Autism Protocols" Face Challenges in Blindness
Most clinical "gold standard" instruments for ASD assessment rely on vision-dependent behaviors (gaze, facial expression, visual social referencing, joint attention). In blindness and severe visual impairment, this leads to a structural impossibility to interpret certain indicators and increases the risk of erroneous conclusions.
A recent scoping review established that the literature on ASD assessment in children with visual impairment/blindness is limited (only 13 empirical articles meeting review criteria) and that issues remain regarding: (a) the suitability of standard instruments, (b) their modifications, (c) the need for new tools, and (d) the need for specialist training (Stevenson & Tedone, 2025).
A significant step forward is the validation of a modified ADOS-2 (Module 3) for children with moderate/severe to profound visual impairment from congenital peripheral disorders: a study with a total of 100 children (aged 4–7), of whom 83 were "verbally fluent," reported good reliability/construct validity and a new "VI algorithm" with high sensitivity and specificity compared to clinical formulation, as well as cut-off values for "Low risk" and "High risk" groups (Dale et al., 2024).
Practical Framework for Differential Diagnosis (Proposal)
In clinical practice, the following reasoning sequence can be used:
- Etiology and Functional Vision: Peripheral impairment versus CVI; degree of functional vision and reliability of visual input.
- Social Motivation Beyond Vision: Seeking contact via voice/touch; response to social proximity; ability to share attention via auditory and tactile means.
- Function of Stereotypies: Self-calibration/self-regulation versus rigid rituals with limited social flexibility.
- History of Early Screen Exposure: Age of onset, intensity, context (feeding/calming before a screen), observed regressions upon increasing or decreasing screen time.
- In Cases of Significant Screen History: Consideration of screening for proposed markers (SIPVR, SIPECR) and assessment for cue-dependent behavior (as hypotheses), parallel to standard ASD assessment.
This framework does not replace diagnostic criteria but aims to reduce the risk of confounding deprivation, disintegration, and primary neurodevelopmental factors. To address "blindness and autism" correctly, diagnosis must be treated as a distinct methodological problem, not merely as "applying the same criteria." It is critical to avoid identical interpretation of stereotypies and rituals, for example – given different mechanisms, they may serve different functions and require different therapeutic approaches.
Practical Implications for Support
- Assessment of auditory-tactile social participation in blind children (how they seek contact, how they share attention without gaze).
- Profiling of play: whether impoverished symbolization is surmountable with scaffolding/verbal and tactile support (more characteristic of VI) or is more resistant and combined with limited social motivation (more characteristic of ASD, though variable) (Bishop et al., 2005).
- Use of adapted instruments and multidisciplinary teams when ASD is suspected in VI/blindness (Stevenson & Tedone, 2025).
- Consideration of etiology: in ONH, ROP, LCA, and other subgroups, the risk of additional neurodevelopmental difficulties may be higher, necessitating planned follow-up and early intervention (de Verdier, Kim, 2018).
Limitations and Future Directions
The present review is narrative and incorporates a conceptual model. The main limitations are:
- Significant heterogeneity exists regarding definitions (blindness vs. VI; peripheral vs. cerebral), assessment tools, and age groups (Fazzi et al., 2019).
- There is a scarcity of validated diagnostic protocols and a need for consensus (Stevenson & Tedone, 2025).
Promising directions for future research include: (a) longitudinal studies of social attention in the absence of vision; (b) comparable paradigms for play and pragmatics; (c) neurodevelopmental mechanisms in "ocular-plus" etiologies (Begeer et al., 2014); (d) experimental paradigms for sensory re-weighting and postural control in ASD, CVI, and blindness; (e) objective vestibular assessments (including nystagmus measurement) in the context of rotational and passive challenges; and (f) validation of the proposed markers (SIPVR/SIPECR) and their specificity across different groups.
Conclusion
The comparison between early blindness and Autism Spectrum Disorder (ASD) reveals a fundamental paradox in developmental neuroscience: distinct sensory etiologies can converge into a strikingly similar behavioral phenotype. While both groups display stereotypies, social withdrawal, and spatial insecurity, this review argues that these behaviors spring from opposing sensory realities. In blindness, the core deficit is sensory deprivation, leading to a compensatory re-weighting toward vestibular and somatosensory inputs; here, rhythmic motor behaviors often serve as functional "self-calibration" in the absence of a visual horizon.
In contrast, the proposed model for screen-induced ASD suggests a mechanism of sensory disintegration. In these cases, vision is not absent but pathologically hyper-dominant and "2D-locked." This artificial visual dominance actively suppresses vestibular and proprioceptive feedback, creating a "sensory mismatch" wherein the brain relies on a static visual anchor that conflicts with physical gravity. The emergence of pathological reflexes such as SIPVR and SIPECR, alongside the absence of post-rotatory nystagmus, supports the hypothesis that the vestibular system is not merely "ignored" but actively inhibited. In this phenotype, the visual system monopolizes cortical resources, resulting in distinct pathological markers:
- Suppression of body-based senses, evidenced by the lack of physiological nystagmus and the onset of panic upon inversion (SIPVR).
- Visual addiction, evidenced by extreme distress and behavioral dysregulation upon eye covering (SIPECR).
Recognizing these markers is critical. While "blindisms" in visually impaired children are compensatory and often transient, the rigid motor rituals in screen-induced ASD represent a blockade of multisensory development. Future clinical protocols should incorporate specific vestibular and sensory-integration challenges to differentiate these groups. Ultimately, understanding these mechanisms offers hope: while deprivation requires lifelong adaptation, disintegration driven by environmental factors may be reversible through therapeutic re-integration of the vestibular and proprioceptive systems.
Therefore, the diagnostic process must move beyond symptom checklists to a functional analysis of the Vision-Vestibular-Proprioception Triad. A "blind-like" behavior in a sighted child should trigger an investigation into sensory integration efficacy and screen history rather than an automatic ASD diagnosis. By distinguishing between the absence of social cues (blindness) and the inability to integrate them (sensory conflict), clinicians can move from generic management to targeted interventions that address the specific sensory architecture of the developing child.
References
American Psychiatric Association. (2013). Diagnostic and statistical manual of mental disorders (5th ed.). (5th ed.). American Psychiatric Association Publishing. https://doi.org/10.1176/appi.books.9780890425596
Arias, P., Bellmann, C., & Aucouturier, J.-J. (2021). Facial mimicry in the congenitally blind. Current Biology, 31(19), R1112–R1114. https://doi.org/10.1016/j.cub.2021.08.059
Begeer, S., Dik, M., Voor De Wind, M. J., Asbrock, D., Brambring, M., & Kef, S. (2014). A New Look at Theory of Mind in Children with Ocular and Ocular-Plus Congenital Blindness. Journal of Visual Impairment & Blindness, 108(1), 17–27. https://doi.org/10.1177/0145482X1410800103
Bishop, M., Hobson, R. P., & Lee, A. (2005). Symbolic play in congenitally blind children. Development and Psychopathology, 17(02). https://doi.org/10.1017/S0954579405050212
Chisari, D., Vitkovic, J., Clark, R., & Rance, G. (2024). Vestibular Function and Postural Control in Children with Autism Spectrum Disorder. Journal of Clinical Medicine, 13(17), 5323. https://doi.org/10.3390/jcm13175323
Chung, S., & Son, J.-W. (2020). Visual Perception in Autism Spectrum Disorder: A Review of Neuroimaging Studies. Journal of the Korean Academy of Child and Adolescent Psychiatry, 31(3), 105–120. https://doi.org/10.5765/jkacap.200018
Dale, N., Sakkalou, E., Eriksson, M. H., & Salt, A. (2024). Modification and Validation of an Autism Observational Assessment Including ADOS-2® for Use with Children with Visual Impairment. Journal of Autism and Developmental Disorders. https://doi.org/10.1007/s10803-024-06514-z
de Verdier, Kim. (2018). Children with blindness: Developmental aspects, comorbidity and implications for education and support [Doctoral thesis, Department of Special Education, Stockholm University]. https://su.diva-portal.org/smash/record.jsf?pid=diva2:1206093&dswid=-684
Fazzi, E., Lanners, J., Danova, S., Ferrarri-Ginevra, O., Gheza, C., Luparia, A., Balottin, U., & Lanzi, G. (1999). Stereotyped behaviours in blind children. Brain and Development, 21(8), 522–528. https://doi.org/10.1016/S0387-7604(99)00059-5
Fazzi, E., Micheletti, S., Galli, J., Rossi, A., Gitti, F., & Molinaro, A. (2019). Autism in Children With Cerebral and Peripheral Visual Impairment: Fact or Artifact? Seminars in Pediatric Neurology, 31, 57–67. https://doi.org/10.1016/j.spen.2019.05.008
Federici, S., Bardin, A., Borsini, C., Delvecchio, E., Lepri, A., Morelli, F., Scognamillo, I., Cocchi, E., Santini, L., & Signorini, S. (2025). Pretend play in children with a congenital visual impairment. Frontiers in Psychology, 16, 1535086. https://doi.org/10.3389/fpsyg.2025.1535086
Helena, R. C. S., Borges, V. M. S., & Sleifer, P. (2025). Postural balance in children and adolescents with Autism Spectrum Disorder: A systematic review. Audiology - Communication Research, 30, e2945. https://doi.org/10.1590/2317-6431-2024-2945en
Manolova, V. R., & Vezenkov, S. R. (2025). Unified Trauma-Addiction Functioning Model. Nootism, 1(4), 4–24. https://doi.org/10.64441/nootism.1.4.1
Manolova, V., & Vezenkov, S. (2025a). Are Screen-Induced Synesthesia and Screen-induced Cue-Dependent Behavior Contributing Factors in the Misdiagnosis of ASD Among Children with Screen Addiction? Nootism, 1, 11–16. https://doi.org/10.64441/nootism.1.3.2
Manolova, V., & Vezenkov, S. (2025b). Screen Trauma – Specifics of the Disorder and Therapy in Adults and Children. Nootism, 1(1), 37–51. https://doi.org/10.64441/nootism.2NCSC.3
Mansour, Y., Burchell, A., & Kulesza, R. J. (2021). Central Auditory and Vestibular Dysfunction Are Key Features of Autism Spectrum Disorder. Frontiers in Integrative Neuroscience, 15, 743561. https://doi.org/10.3389/fnint.2021.743561
Manzari, L., Perez-Fernandez, N., & Tramontano, M. (2022). Editorial: Gaze and postural stability rehabilitation. Frontiers in Neurology, 13, 1034012. https://doi.org/10.3389/fneur.2022.1034012
Nomi, J. S., & Uddin, L. Q. (2015). Face processing in autism spectrum disorders: From brain regions to brain networks. Neuropsychologia, 71, 201–216. https://doi.org/10.1016/j.neuropsychologia.2015.03.029
Spiteri, S., & Crewther, D. (2021). Neural Mechanisms of Visual Motion Anomalies in Autism: A Two-Decade Update and Novel Aetiology. Frontiers in Neuroscience, 15, 756841. https://doi.org/10.3389/fnins.2021.756841
Stevenson, M., & Tedone, E. (2025). Assessment of Autism Spectrum Disorders in Children with Visual Impairment and Blindness: A Scoping Review. Journal of Autism and Developmental Disorders, 55(4), 1411–1423. https://doi.org/10.1007/s10803-024-06300-x
Tadić, V., Pring, L., & Dale, N. (2010). Are language and social communication intact in children with congenital visual impairment at school age? Journal of Child Psychology and Psychiatry, 51(6), 696–705. https://doi.org/10.1111/j.1469-7610.2009.02200.x
Vezenkov, S., & Manolova, V. (2025a). Screen-Induced Pathological Eye-Covering Reflex in Children with Early Screen Addiction. Nootism, 1(3), 5–10. https://doi.org/10.64441/nootism.1.3.1
Vezenkov, S., & Manolova, V. (2025b). Screen-Induced Pathological Vestibular Reflex: A Specific Marker of Early Screen Addiction. Nootism, 1(2), 5–10. https://doi.org/10.64441/nootism.1.2.1
Vezenkov, S. R., & Manolova, V. R. (2025). Neurobiology of Autism/Early Screen Addiction Recovery. Nootism, 1, 19–36. https://doi.org/10.64441/nootism.2NCSC.2
Zhang, C., Lee, T. M. C., Fu, Y., Ren, C., Chan, C. C. H., & Tao, Q. (2019). Properties of cross-modal occipital responses in early blindness: An ALE meta-analysis. NeuroImage: Clinical, 24, 102041. https://doi.org/10.1016/j.nicl.2019.102041
Zilbovicius, M., Meresse, I., Chabane, N., Brunelle, F., Samson, Y., & Boddaert, N. (2006). Autism, the superior temporal sulcus and social perception. Trends in Neurosciences, 29(7), 359–366. https://doi.org/10.1016/j.tins.2006.06.004